study visit checklist ensure consistency and documentation of study visits. principal investigator: ____________________ irb#: ________
STUDY VISIT CHECKLIST
Ensure consistency and documentation of study visits.
Principal Investigator: ____________________ IRB#:
______________________ Sponsor: _________________________
Study Title:
_______________________________________________________________________________________________
SUBJECT ID: DOB:
INFORMED CONSENT
================
PI/Authorized Staff Explained Study
PI/Staff:
Copy of consent given to adult subject and/or LAR; child subject
and/or Parent or Legal Guardian
Adult Subject and/or LAR; Child Subject and/or Parent or Legal
Guardian- Signed Consent
Date Signed:
Is Consent Valid?
YES NO
If subject did not sign consent, explain:
STUDY VISITS
**Please customize this form to meet the visit requirements of your
specific study.
Study Visit 1:
Date Completed:
PI/Staff Initials
If subject did not complete test or completed test on different date,
please explain:
e.g. Complete Blood Count
e.g. Pulmonary Function Test
e.g. EKG
e.g. Chest x-ray
Study Visit 2:
Date Completed:
PI/Staff Initials
If subject did not complete test or completed test on different date,
please explain:
Study Completion:
If subject did not complete study, please explain:
Subject Completed Study
Date Completed:
If applicable, study reimbursement
Date Given:
NOTES:
Page _____ of _____
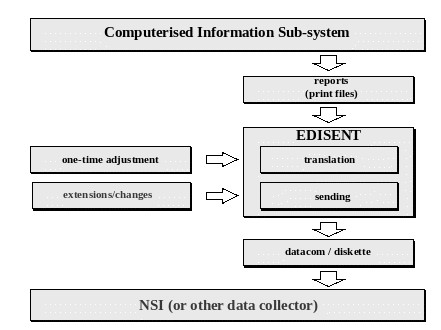 11 SESSIONNO 2 PAPER NO 5 COUNTRY ITALY DEVELOPMENT
11 SESSIONNO 2 PAPER NO 5 COUNTRY ITALY DEVELOPMENT Boards-of-Examiners-and-the-Academic-Progress-Review-Sub-Committee-Policy
Boards-of-Examiners-and-the-Academic-Progress-Review-Sub-Committee-Policy RECETA DEL LIBRO EL LIBRO DEL SABER CULINARIO FORMULACIÓN
RECETA DEL LIBRO EL LIBRO DEL SABER CULINARIO FORMULACIÓN C ONCURSO LITERARIO NACIONAL “SERAFÍN J GARCÍA” DÉCIMO QUINTA
C ONCURSO LITERARIO NACIONAL “SERAFÍN J GARCÍA” DÉCIMO QUINTA 20 DE ABRIL – 3 DE JUNIO DE
20 DE ABRIL – 3 DE JUNIO DE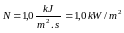 SLOVENSKÝ VÝBOR FYZIKÁLNEJ OLYMPIÁDY TR A HLINKU 1 949
SLOVENSKÝ VÝBOR FYZIKÁLNEJ OLYMPIÁDY TR A HLINKU 1 949 „SENSORINIO GESINTUVO“ RINKINYS LIETUVOS BIBLIOTEKOMS (ATMINTINĖ) ASS TURINTIEMS ASMENIMS
„SENSORINIO GESINTUVO“ RINKINYS LIETUVOS BIBLIOTEKOMS (ATMINTINĖ) ASS TURINTIEMS ASMENIMS Guia Docente de la Asignatura (∾) Nombre de la
Guia Docente de la Asignatura (∾) Nombre de la NAME CLASS DATE ACTIVITY B07 MEMBRANE
NAME CLASS DATE ACTIVITY B07 MEMBRANE “2013 AÑO DEL BICENTENARIO DE LA ASAMBLEA G
“2013 AÑO DEL BICENTENARIO DE LA ASAMBLEA G