sop: standard operating procedures number date author approved by page hrp-071 6/8/2010 c. bienstock j. si
SOP: Standard Operating Procedures
NUMBER
DATE
AUTHOR
APPROVED BY
PAGE
HRP-071
6/8/2010
C. Bienstock
J. Silverstein
1 of 1
1.
PURPOSE
1.
This procedure establishes the process to create and update
standard operating procedures and associated checklists and
worksheets.
2.
The process begins when the IRB manager or Organizational
Official determines that a standard operating procedure needs
to be created or modified.
3.
The process ends when the new or revised standard operating
procedure has been approved and filed.
2.
REVISIONS FROM PREVIOUS VERSION
1.
None
3.
POLICY
1.
None
4.
RESPONSIBILITIES
1.
The IRB manager carries out these procedures.
5.
PROCEDURE
1.
For new standard operating procedure, assign a number.
2.
Assign an author and approver.
3.
Have the author create or update the standard operating
procedure following the “TEMPLATE SOP” or update the
associated checklist or worksheet.
4.
Have the approver review and approve the document.
5.
Once approved by the approver:
1.
Update the approval date.
2.
File the approved new or revised document in the standard
operating procedure files.
3.
Post the approved procedure on the Human Research
Protection Program Web site.
4.
File the old document, if any, in the standard operating
procedure files.
5.
Send an email to affected individuals informing them of
the change.
6.
MATERIALS
1.
TEMPLATE SOP
7.
REFERENCES
1.
None
 CREAR UN MAPA CONCEPTUAL CON CMAPTOOLS A CONTINUACIÓN EJEMPLIFICAREMOS
CREAR UN MAPA CONCEPTUAL CON CMAPTOOLS A CONTINUACIÓN EJEMPLIFICAREMOS MODELO DE SOLICITUD DE PARTICIPACIÓN EN LA ACTUACIÓN MISIÓN
MODELO DE SOLICITUD DE PARTICIPACIÓN EN LA ACTUACIÓN MISIÓN 2015 ČRNA NA KOROŠKEM 30 JANUAR DO 1FEBRUAR SPOŠTOVANI
2015 ČRNA NA KOROŠKEM 30 JANUAR DO 1FEBRUAR SPOŠTOVANI RESUMES & COVER LETTERS DEPENDING ON WHOM YOU ASK
RESUMES & COVER LETTERS DEPENDING ON WHOM YOU ASK AHRC2827 UNITED NATIONS AHRC2827 GENERAL ASSEMBLY DISTR GENERAL 17
AHRC2827 UNITED NATIONS AHRC2827 GENERAL ASSEMBLY DISTR GENERAL 17 DURINGAFTER READING – ANALYZE & RESPOND JIGSAW WHAT? A
DURINGAFTER READING – ANALYZE & RESPOND JIGSAW WHAT? A TERCER LABORATORIO DE PROPUESTAS Y PRESENTACIÓN DE PROYECTOS DE
TERCER LABORATORIO DE PROPUESTAS Y PRESENTACIÓN DE PROYECTOS DE ORSZÁGOS EGÉSZSÉGBIZTOSÍTÁSI PÉNZTÁR IGÉNYLŐLAP EURÓPAI EGÉSZSÉGBIZTOSÍTÁSI KÁRTYÁHOZ MELY AZ
ORSZÁGOS EGÉSZSÉGBIZTOSÍTÁSI PÉNZTÁR IGÉNYLŐLAP EURÓPAI EGÉSZSÉGBIZTOSÍTÁSI KÁRTYÁHOZ MELY AZ PQ002 PAGE 1 SUR 1 DATE CRÉATION 21112018 MODIFICATION
PQ002 PAGE 1 SUR 1 DATE CRÉATION 21112018 MODIFICATION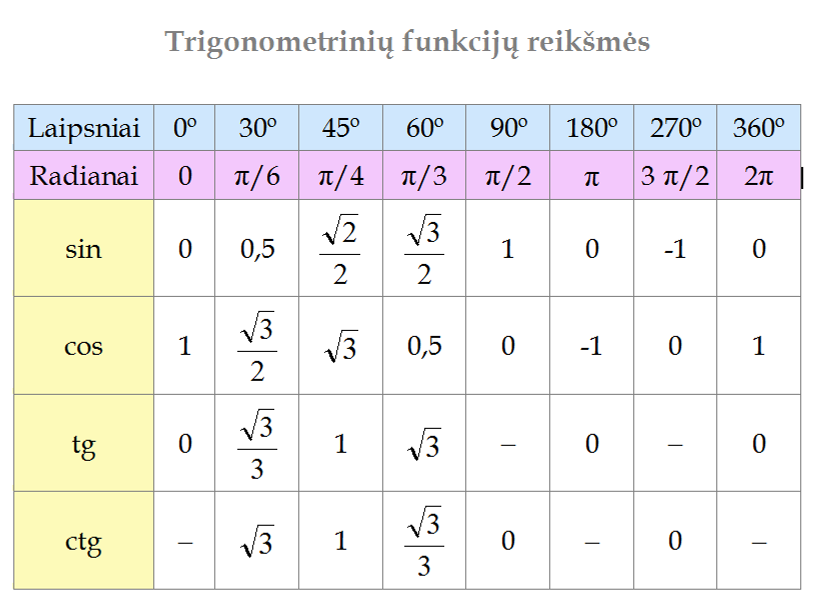 AUTOMATINIS LENTELIŲ FORMATAVIMAS LENTELĖS DUOMENŲ RIKIAVIMAS 1 UŽDUOTIS ŽEMIAU
AUTOMATINIS LENTELIŲ FORMATAVIMAS LENTELĖS DUOMENŲ RIKIAVIMAS 1 UŽDUOTIS ŽEMIAU STAFF NURSE III AND HHH III RENEWAL PACKET A
STAFF NURSE III AND HHH III RENEWAL PACKET A NAGRADNI NATEČAJ REVIJE TIM IN LEPIL UHU UREDNIŠTVO
NAGRADNI NATEČAJ REVIJE TIM IN LEPIL UHU UREDNIŠTVO