institutional animal care & use committee ========================================= 270 engelmann hall p. o. box 413 milwaukee
Institutional Animal Care & Use Committee
=========================================
270 Engelmann Hall
P. O. Box 413
Milwaukee, WI
53201-0413
(414) 229-6339 phone
(414) 229-6729 fax
www.uwm.edu/animal-care
IACUC Guidelines SOP for Sterile Preparation of Injectable Compounds
The syringe, filter and receiving container are all sterile. Also,
while not necessary, doing the procedure in a biosafety hood is
recommended to improve sterile transfer.
Procedures:
1.
If you have a limited amount of sample – draw a small amount of
air (about 1 ml) into a sterile syringe before filling with the
sample solution. This air is used to purge the filter at the end
ensuring the minimum sample left in the filter.
2.
Load the sample into the syringe. Note the visible air pocket
ready to purge the filter.
3.
Attach the sterile filter securely with a twisting motion. Filters
with nominal pore size of 0.2 micron are typical for sterilizing
applications (but 0.1 and 0.45 are also common). With a luer slip
syringe, this is about one quarter turn as the filter is pushed
on. If the syringe has a luer lock (as in this example), fix it
firmly but do not over-tighten.
4.
Hold the assembled syringe and the filter vertically to wet the
membrane evenly. This prevents air blocks and promotes high flow
rates as the sample is spread evenly over the membrane surface.
The actual physical design of the syringe filter will have a big
bearing on the possible flow-rate through the filter.
5.
Press the syringe plunger gently to express the sample through the
filter. If the back pressure ever increases significantly, change
the filter as it may have plugged. Avoid pressing excessively as
this could cause the filter housing to burst.
6.
Push the air through the filter to purge the housing and membrane
and recover the maximum amount of sample.
7.
In all steps use aseptic technique.
NOTE: There are many manufacturers of syringe filters (e.g., Fisher)
so PI’s should be able to find the membrane material to fit their
needs. They may cost about $2.00 each and are single use. If there is
concern about filter hold up of the active substance it could be
confirmed by any analytical method suitable for the active substance
(UV/Vis, MS, Elisa) whereby the concentrations in the pre-and
post-filtered substance are compared.
Sterile is required in addition to pharmaceutical grade (note that
pharmaceutical grade does not necessarily mean sterile) for anything
that is injected if/when practical sterilization methods (as above)
are available unless a scientific justification is provided to the
IACUC and approved.
Reviewed: 4/29/2016
Page 2
 SECTOR SUBSECTOR DE APRENDIZAJE ESPECIALIDAD LENGUA Y LITERATURA CURSO(S)
SECTOR SUBSECTOR DE APRENDIZAJE ESPECIALIDAD LENGUA Y LITERATURA CURSO(S) ANEXO V CONSEJERÍA DE EDUCACIÓN FORMACIÓN PROFESIONAL Y TURISMO
ANEXO V CONSEJERÍA DE EDUCACIÓN FORMACIÓN PROFESIONAL Y TURISMO TEGEVUSTOETUSE 2013 AASTA ARUANNE RIIGIEELARVELISE ERALDISE KASUTAMISE TÄITMISE ARUANNE
TEGEVUSTOETUSE 2013 AASTA ARUANNE RIIGIEELARVELISE ERALDISE KASUTAMISE TÄITMISE ARUANNE VECTOR CONCEPTS FOR FLUID MECHANICS 1 A VECTOR HAS
VECTOR CONCEPTS FOR FLUID MECHANICS 1 A VECTOR HAS SERVICIO DE DEPORTES PROTOCOLO COVID19 INICIO ACTIVIDADES DEPORTIVAS 2021
SERVICIO DE DEPORTES PROTOCOLO COVID19 INICIO ACTIVIDADES DEPORTIVAS 2021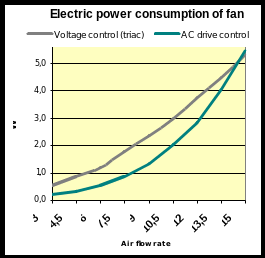 4 CONVERTITORI DI FREQUENZA NELLE CENTRALI DI REFRIGERAZIONE SILVIO
4 CONVERTITORI DI FREQUENZA NELLE CENTRALI DI REFRIGERAZIONE SILVIO CONSEJERÍA DE DESARROLLO SOSTENIBLE VICECONSEJERÍA DE MEDIO AMBIENTE MJAT
CONSEJERÍA DE DESARROLLO SOSTENIBLE VICECONSEJERÍA DE MEDIO AMBIENTE MJAT CONTACT JUDY IANNACCONE DIRECTOR COMMUNICATIONS JANUARY 28 2008 PHONE
CONTACT JUDY IANNACCONE DIRECTOR COMMUNICATIONS JANUARY 28 2008 PHONE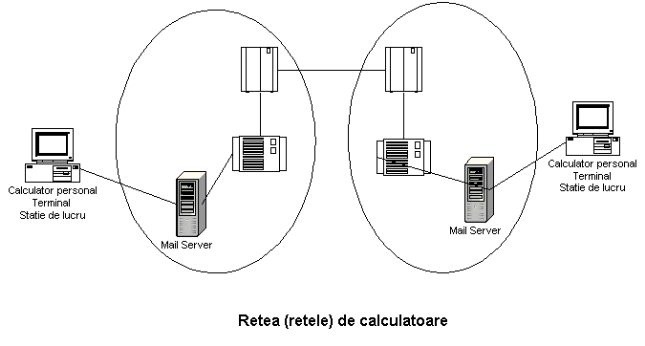 GHIDUL UTILIZATORULUI CUTIEI POȘTALE ELECTRONICE ÎN CADRUL INSTITUȚIILOR AȘM
GHIDUL UTILIZATORULUI CUTIEI POȘTALE ELECTRONICE ÎN CADRUL INSTITUȚIILOR AȘM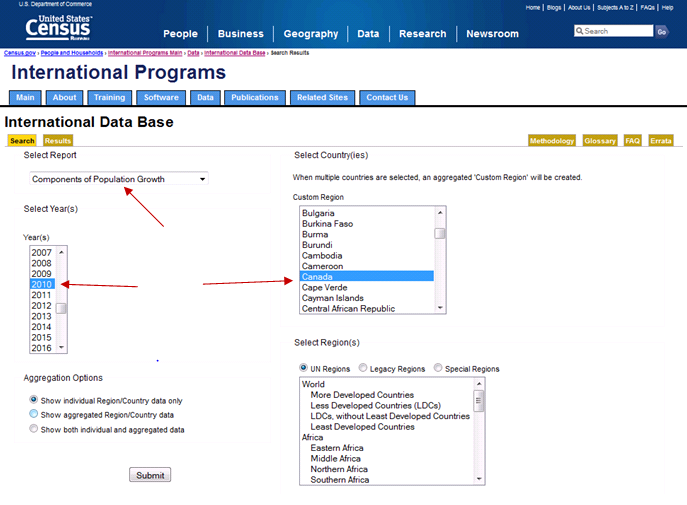 ALL USERS CHAPTER 7 PROJECT HUMAN POPULATION AND MIGRATION
ALL USERS CHAPTER 7 PROJECT HUMAN POPULATION AND MIGRATION